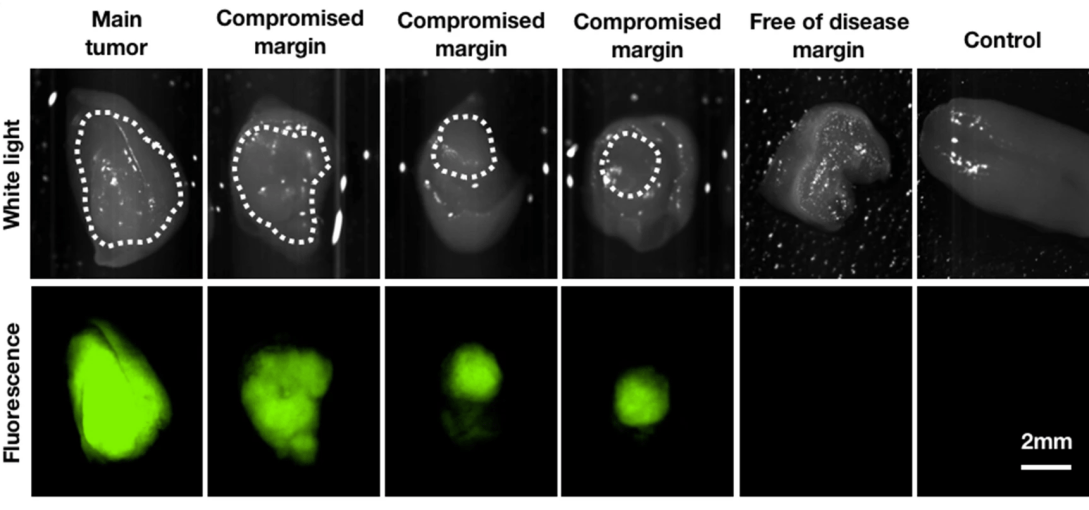
Complete removal and negative margins are the goal of any surgical resection of primary oral cavity carcinoma. Current approaches to determine tumor boundaries rely heavily on surgeons’ expertise, and final histopathological reports are usually only available days after surgery, precluding contemporaneous re-assessment of positive margins. Intraoperative optical imaging could address this unmet clinical need. Using mouse models of oral cavity carcinoma, we demonstrated that PARPi-FL, a fluorescent PARP inhibitor targeting the enzyme PARP1/2, can delineate oral cancer and accurately identify positive margins, both macroscopically and at cellular resolution. PARPi-FL also allowed identification of compromised margins based on fluorescence hotspots, which were not seen in margin-negative resections and control tongues. PARPi-FL was further able to differentiate tumor from low-grade dysplasia. Intravenous injection of PARPi-FL has significant potential for clinical translation and could aid surgeons in assessing oral cancer margins in vivo.
Author: Sheryl Roberts
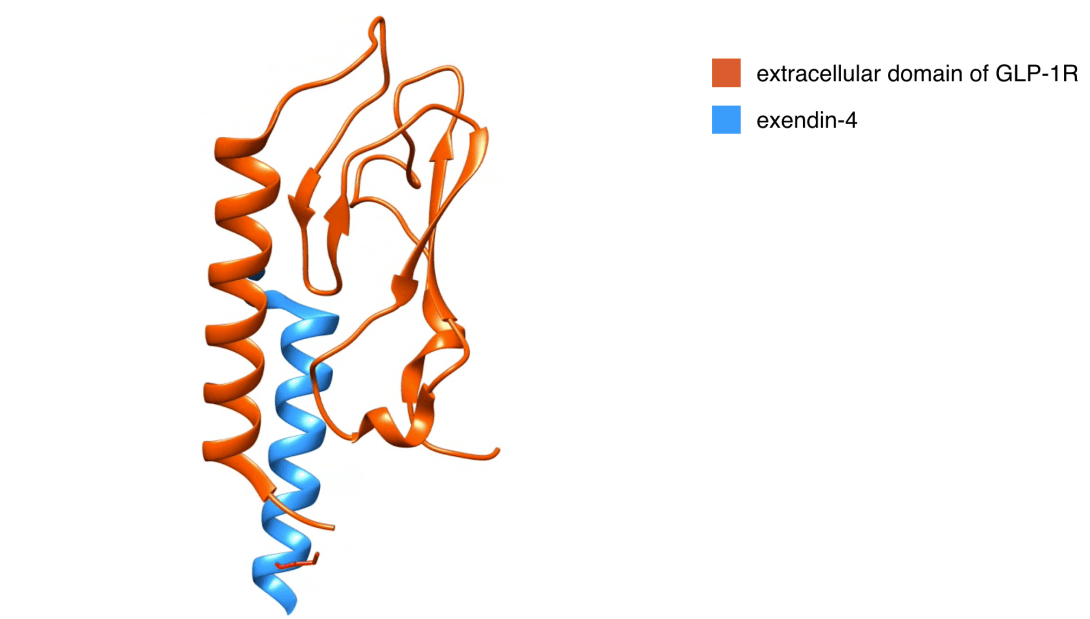
Optoacoustic imaging of GLP-1 Receptor with a near-infrared exendin-4 analog
Limitations in current imaging tools have long challenged the imaging of small pancreatic islets in animal models. Here, we report the first development and in vivo validation testing of a broad spectrum and high absorbance near infrared optoacoustic contrast agent, E4x12-Cy7. Our near infrared tracer (E4x12-Cy7) is based on the amino acid sequence of exendin-4 and targets the glucagon-like peptide-1 receptor (GLP-1R). Cell assays confirmed that E4x12-Cy7 has a high binding affinity (IC50 = 4.6 ± 0.8 nM). Using the multi-spectral optoacoustic tomography (MSOT), we imaged E4x12-Cy7 and optoacoustically visualized ß-cell insulinoma xenografts in vivo for the first time. In the future, similar optoacoustic tracers that are specific for ß-cells and combines optoacoustic and fluorescence imaging modalities could prove to be important tools for monitoring the pancreas for the progression of diabetes.
Safety and feasibility of PARP1/2 imaging with 18F-PARPi in patients with head and neck cancer
Purpose. We performed a first-in-human clinical trial. The aim of this study was to determine safety and feasibility of PET imaging with 18F-PARPi in patients with head and neck cancer. Patients and Methods. Eleven patients with newly diagnosed or recurrent oral and oropharyngeal cancer were injected with 18F-PARPi (331 ± 42 MBq) and dynamic PET/CT imaging was performed between 0 min and 25 min post-injection. Static PET/CT scans were obtained at 30 min, 60 min and 120 min post injection. Blood samples for tracer concentration and metabolite analysis were collected. Blood pressure, ECG, oxygen levels, clinical chemistry and CBC were obtained before and after tracer administration.
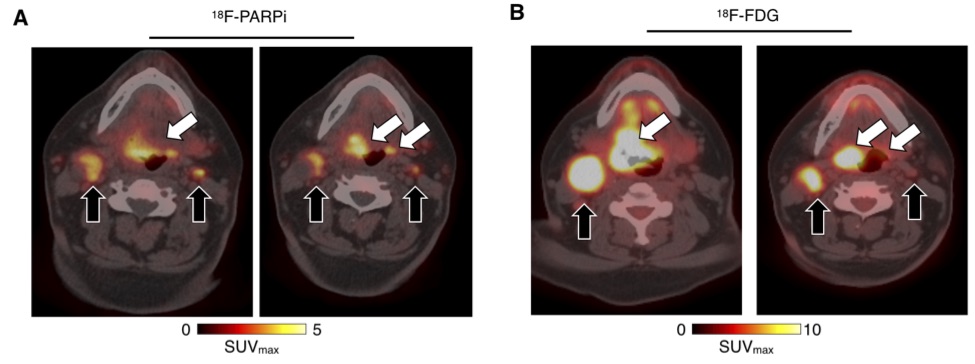
Results. 18F-PARPi was well-tolerated by all patients without any safety concerns. Of the 11 patients included in the analysis, 18F-PARPi had focal uptake in all primary lesions (n = 10, SUVmax = 2.8 ± 1.2) and all 18F-FDG positive lymph nodes (n = 34). 18F-PARPi uptake was seen in 18F-FDG negative lymph nodes of three patients (n = 6). Focal uptake of tracer in primary and metastatic lesions was corroborated by CT alone or in combination with 18F-FDG. The overall equivalent dose with 18F-PARPi PET was 3.9 mSv – 5.2 mSv, contrast was high (SUVmax(lesion)/SUVmax(trapezius muscle) = 4.5) and less variable than 18F-FDG when compared to the genioglossus muscle (1.3 versus 6.0, p = 0.001).
Conclusions. Imaging of head and neck cancer with 18F-PARPi is feasible and safe. 18F-PARPi detects primary and metastatic lesions, and retention in tumors is longer than in healthy tissues.
High-resolution optoacoustic imaging of tissue responses to vascular-targeted therapies
The monitoring of vascular-targeted therapies using magnetic resonance imaging, computed tomography or ultrasound is limited by their insufficient spatial resolution. Here, by taking advantage of the intrinsic optical properties of haemoglobin, we show that raster-scanning optoacoustic mesoscopy (RSOM) provides high-resolution images of the tumour vasculature and of the surrounding tissue, and that the detection of a wide range of ultrasound bandwidths enables the distinction of vessels of differing size, providing detailed insights into the vascular responses to vascular-targeted therapy. Using RSOM to examine the responses to vascular-targeted photodynamic therapy in mice with subcutaneous xenografts, we observed a substantial and immediate occlusion of the tumour vessels followed by haemorrhage within the tissue and the eventual collapse of the entire vasculature. Using dual-wavelength RSOM, which distinguishes oxyhaemoglobin from deoxyhaemoglobin, we observed an increase in oxygenation of the entire tumour volume immediately after the application of the therapy, and a second wave of oxygen reperfusion approximately 24 h thereafter. We also show that RSOM enables the quantification of differences in neoangiogenesis that predict treatment efficacy.
18F-Galactodendritic Carbohydrate for Bladder Cancer PET/CT
# Equally contributed
Galectins are carbohydrate-binding proteins overexpressed in bladder cancer (BCa) cells. Dendritic galactose moieties have a high affinity for galectin-expressing tumor cells. We radiolabeled a dendritic galactose carbohydrate with fluorine-18 – 18F-labeled galactodendritic unit 4 – and examined its potential in imaging urothelial malignancies. Methods: The 18F-labeled 1st generation galactodendritic unit 4 was obtained from its tosylate precursor. We conducted in vivo studies in galectin-expressing UMUC3 orthotopic BCa model to determine the ability of 18F-labeled galactodendritic unit 4 to image BCa. Results: Intravesical administration of 18F-labeled galactodendritic unit 4 allowed specific accumulation of the carbohydrate radiotracer in galectin-1 overexpressing UMUC3 orthotopic tumors when imaged with PET. The 18F-labeled galactodendritic unit 4 was not found to accumulate in non-tumor murine bladders. Conclusion: The 18F-labeled galactodendritic unit 4 and similar analogs may be clinically relevant and exploitable for PET imaging of galectin-1 overexpressing bladder tumors.
Fluorine-18 labeled PARP inhibitor as a potential alternative to 2-deoxy-2-[18F]fluoro-d-glucose PET in oral cancer imaging
Objectives: The evaluation of disease extent and post-therapy surveillance of head and neck cancer using 2-deoxy-2-[18F]fluoro-d-glucose ([18F]FDG) PET is often complicated by physiological uptake in normal tissues of the head and neck region, especially after surgery or radiotherapy. However, irrespective of low positive predictive values, [18F]FDG PET remains the standard of care to stage the disease and monitor recurrences. Here, we report the preclinical use of a targeted poly (ADP-ribose) polymerase1 (PARP1) binding PET tracer, fluorine-18 labeled poly (ADP-ribose) polymerase1 inhibitor ([18F]PARPi), as a potential alternative with greater specificity.
Methods: Using an orthotopic xenograft mouse model injected with either FaDu or Cal 27 (human squamous cell carcinoma cell lines) we performed PET/CT scans with the 2 tracers and compared the results. Gamma counts and autoradiography were also assessed and correlated with histology.
Results: The average retained activity of [18F]PARPi across cell lines in tumor-bearing tongues was 0.9 ± 0.3%ID/g, 4.1 times higher than in control (0.2 ± 0.04%ID/g). Autoradiography and histology confirmed that the activity arose almost exclusively from the tumor areas, with a signal/normal tissue around a ratio of 42.9 ± 21.4. In vivo, [18F]PARPi-PET allowed delineation of tumor from healthy tissue (p < .005), whereas [18F]FDG failed to do so (p = .209).
Conclusions and implications for patient care: We demonstrate that [18F]PARPi is more specific to tongue tumor tissue than [18F]FDG. [18F]PARPi PET allows for the straightforward delineation of oral cancer in mouse models, suggesting that clinical translation could result in improved imaging of head and neck cancer when compared to [18F]FDG.
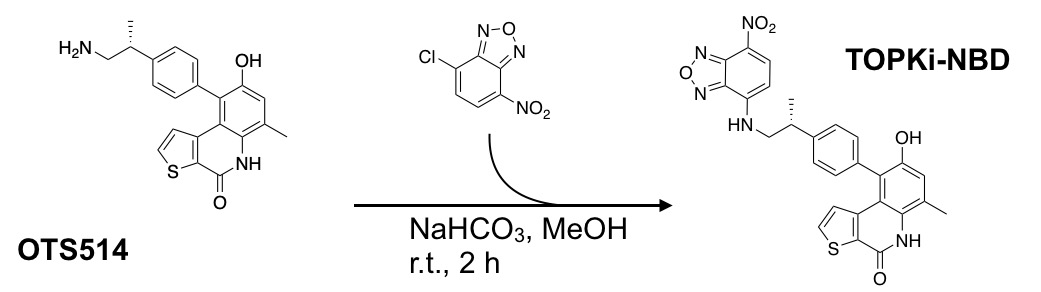
TOPKi-NBD: a fluorescent small molecule for tumor imaging
Giacomo Pirovano, Sheryl Roberts, Thomas Reiner*
Purpose: OTS514 is a highly specific inhibitor targeting lymphokine-activated killer T cell-originated protein kinase (TOPK). A fluorescently labeled TOPK inhibitor could be used for tumor delineation or intraoperative imaging, potentially improving patient care. Methods: Fluorescently labeled OTS514 was obtained by conjugating the fluorescent small molecule NBD to the TOPK inhibitor. HCT116 colorectal cancer cells were used to generate tumors in NSG mice for in vivo studies. Images were generated in vitro using confocal microscopy and ex vivo using an IVIS Spectrum. Results: OTS514 was successfully conjugated to a fluorescent sensor and validated in vitro, in vivo, and ex vivo. The labeling reaction led to TOPKi-NBD with 67% yield and 97% purity after purification. We were able to test binding properties of TOPKi-NBD to its target, TOPK, and compared them to the precursor inhibitor. EC50s showed similar target affinities for TOPKi-NBD and the unlabeled OTS514. TOPKi-NBD showed specific tumor uptake after systemic administration and was microscopically detectable inside cancer cells ex vivo. Blocking controls performed with an excess of the unlabeled OTS514 confirmed specificity of the compound. Overall, the results represent a first step toward the development of a class of TOPK-specific fluorescent inhibitors for in vivo imaging and tumor delineation. Conclusions: TOPK has the potential to be a new molecular target for cancer-specific imaging in a large variety of tumors. This could lead to broad applications in vitro and in vivo.
PARP1/2 imaging with 18F-PARPi in patients with head and neck cancer
Purpose. We performed a first-in-human clinical trial. The aim of this study was to determine safety and feasibility of PET imaging with 18F-PARPi in patients with head and neck cancer. Patients and Methods. Eleven patients (age 49 to 86 years) with newly diagnosed or recurrent oral and oropharyngeal cancer were injected intravenously with 18F-PARPi (331 ± 42 MBq) and dynamic PET/CT imaging was performed between 0 min and 25 min post-injection. Static PET/CT scans were obtained at 30 min, 60 min and 120 min p.i. Blood samples for tracer concentration and metabolite analysis were collected. Blood pressure, ECG, oxygen levels, clinical chemistry and CBC were obtained before and after administration of 18F-PARPi. Results. 18F-PARPi was well-tolerated by all patients without any safety concerns. Of the 11 patients included in the analysis, 18F-PARPi had focal uptake in all primary lesions (n = 10, SUVmax = 2.8 ± 1.2) and all 18F-FDG positive lymph nodes (n = 34). 18F-PARPi uptake was seen in 18F-FDG negative lymph nodes of three patients (n = 6). Focal uptake of tracer in primary and metastatic lesions was corroborated by CT alone or in combination with 18F-FDG. Contrast for 18F-PARPi and 18F-FDG was comparable (SUVmax(lesion)/SUVmax(genioglossus) = 3.3 and 3.0, respectively; p = 0.23), and SUVmax values for 18F-PARPi were less variable compared to 18F-FDG (1.3 versus 6.0, p = 0.001). Conclusions. Imaging of head and neck cancer with 18F-PARPi is feasible and safe. 18F-PARPi detects primary and metastatic lesions, and retention in tumors is longer than in healthy tissues.
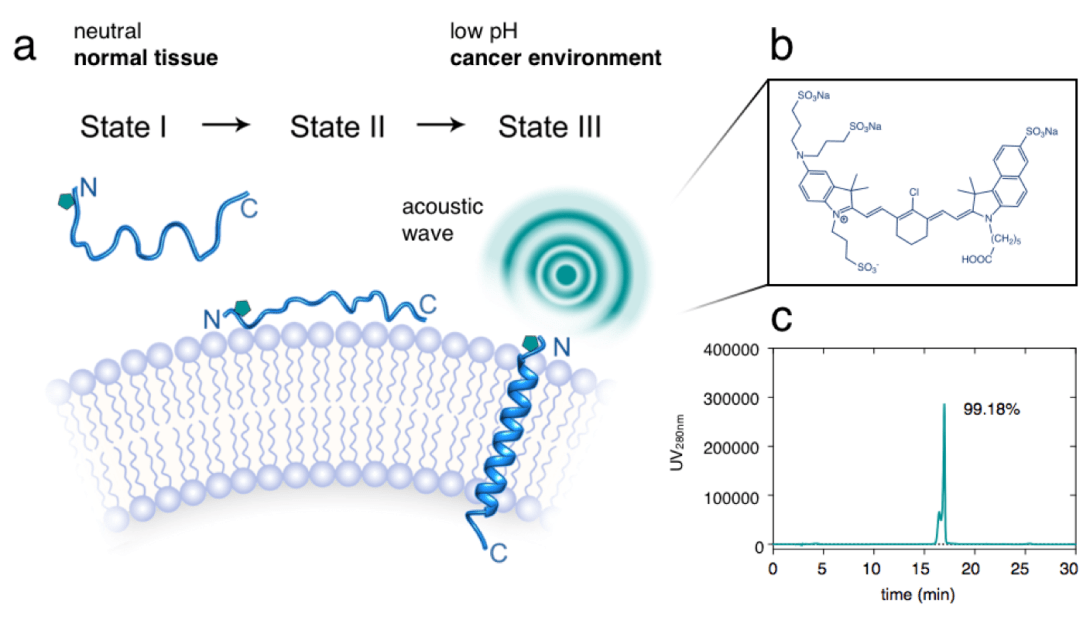
Acid specific dark quencher QC1 pHLIP for multi-spectral optoacoustic diagnoses of breast cancer
Breast cancer is the most common type of malignant growth in women. Early detection of breast cancer, as well as the identification of possible metastatic spread poses a significant challenge because of the structural and genetic heterogeneity that occurs during the progression of the disease. Currently, mammographies, biopsies and MRI scans are the standard of care techniques used for breast cancer diagnosis, all of which have their individual shortfalls, especially when it comes to discriminating tumors and benign growths. With this in mind, we have developed a non-invasive optoacoustic imaging strategy that targets the acidic environment of breast cancer. A pH low insertion peptide (pHLIP) was conjugated to the dark quencher QC1, yielding a non-fluorescent sonophore with high extinction coefficient in the near infrared that increases signal as a function of increasing amounts of membrane insertion. In an orthotopic murine breast cancer model, pHLIP-targeted optoacoustic imaging allowed us to differentiate between healthy and breast cancer tissues with high signal/noise ratios. In vivo, the sonophore QC1-pHLIP could detect malignancies at higher contrast than its fluorescent analog ICG-pHLIP, which was developed for fluorescence-guided surgical applications. PHLIP-type optoacoustic imaging agents in clinical settings are attractive due to their ability to target breast cancer and a wide variety of other malignant growths for diagnostic purposes. Intuitively, these agents could also be used for visualization during surgery.
Blocking Glucagon Like Peptide-1 Receptors in the Exocrine Pancreas Improves Specificity for Beta Cells in a Mouse Model of Type 1 Diabetes
# Equally contributed
The diabetes community has long desired an imaging agent to quantify the number of insulin-secreting beta (β) cells, beyond just functional equivalents (insulin secretion), to help diagnose and monitor early stages of both type 1 and type 2 diabetes mellitus. Loss in the number of β-cells can be masked by a compensatory increase in function of the remaining cells. Since β-cells only form ~1% of the pancreas and decrease as the disease progresses, only a few imaging agents, such as exendin, have demonstrated clinical potential to detect a drop in the already scarce signal. However, clinical translation of imaging with exendin has been hampered by higher than expected pancreatic uptake in subjects with long-term diabetes who lack β-cells. Exendin binds glucagon-like peptide 1 receptor (GLP-1R), previously thought to be expressed only on β-cells, but recent studies report low levels of GLP-1R on exocrine cells, complicating β-cell mass quantification. Here we use a GLP-1R knockout mouse model to demonstrate that exocrine binding of exendin is exclusively via GLP-1R (~1,000/cell), and not any other receptors. We then use lipophilic Cy-7 exendin to selectively pre-block exocrine GLP-1R in healthy and streptozotocin-induced diabetic (STZ) mice. Sufficient receptors remain on β-cells for subsequent labeling with a fluorescent- or 111In-exendin, which improves contrast between healthy and diabetic pancreata, and provides a potential avenue for achieving the long-standing goal of imaging β-cell mass in the clinic.
Nanoemulsion-based delivery of fluorescent PARP inhibitors in mouse models of small cell lung cancer
The preclinical potential of many diagnostic and therapeutic small molecules is limited by their rapid washout kinetics and consequently modest pharmacological performances. In several cases, these could be improved by loading the small molecules into nanoparticulates, improving blood half-life, in vivo uptake and overall pharmacodynamics. In this study, we report a nanoemulsion (NE) encapsulated form of PARPi-FL. As a proof of concept, we used PARPi-FL, which is a fluorescently labeled sensor for olaparib, a FDA-approved small molecule inhibitor of the nuclear enzyme poly(ADP-ribose)polymerase 1 (PARP1). Encapsulated PARPi-FL showed increased blood half-life, and delineated subcutaneous xenografts of small cell lung cancer (SCLC), a fast-progressing disease where efficient treatment options remain an unmet clinical need. Our study demonstrates an effective method for expanding the circulation time of a fluorescent PARP inhibitor, highlighting the pharmacokinetic benefits of nanoemulsions as nanocarriers and confirming the value of PARPi-FL as an imaging agent targeting PARP1 in small cell lung cancer.
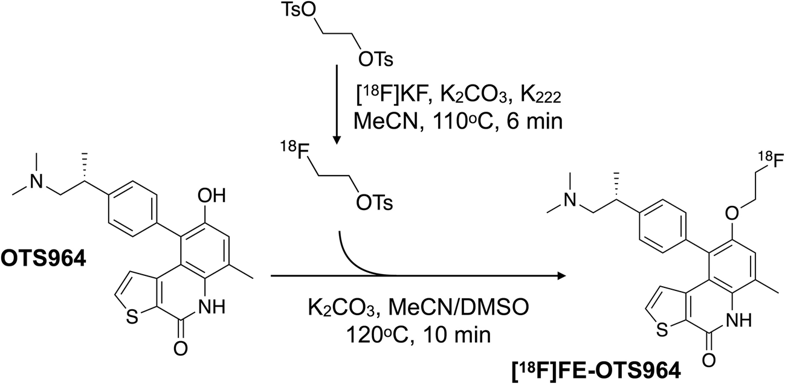
[18F]FE-OTS964: a Small Molecule Targeting TOPK for In Vivo PET Imaging in a Glioblastoma Xenograft Model
Purpose: Lymphokine-activated killer T cell-originated protein kinase (TOPK) is a fairly new cancer biomarker with great potential for clinical applications. The labeling of a TOPK inhibitor with F-18 can be exploited for positron emission tomography (PET) imaging allowing more accurate patient identification, stratification, and disease monitoring.
Procedures: [18F]FE-OTS964 was produced starting from OTS964, a preclinical drug which specifically binds to TOPK, and using a two-step procedure with [18F]fluoroethyl p-toluenesulfonate as a prosthetic group. Tumors were generated in NSG mice by subcutaneous injection of U87 glioblastoma cells. Animals were injected with [18F]FE-OTS964 and PET imaging and ex vivo biodistribution analysis was carried out.
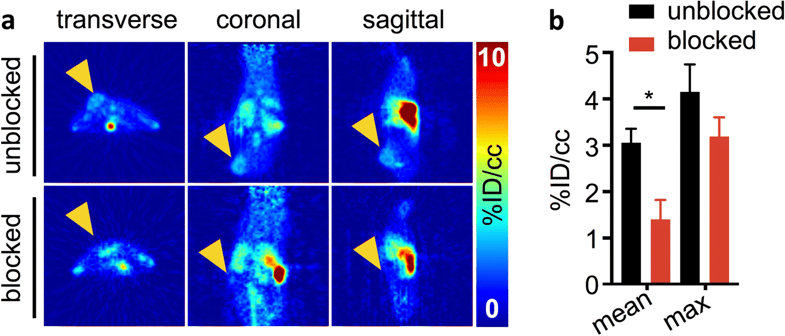
Results: [18F]FE-OTS964 was successfully synthesized and validated in vivo as a PET imaging agent. The labeling reaction led to 15.1 ± 7.5 % radiochemical yield, 99 % radiochemical purity, and high specific activity. Chemical identity of the radiotracer was confirmed by co-elution on an analytical HPLC with a cold-labeled standard. In vivo PET imaging and biodistribution analysis showed tumor uptake of 3.06 ± 0.30 %ID/cc, which was reduced in animals co-injected with excess blocking dose of OTS541 to 1.40 ± 0.42 %ID/cc.
Conclusions: [18F]FE-OTS964 is the first TOPK inhibitor for imaging purposes and may prove useful in the continued investigation of the pharmacology of TOPK inhibitors and the biology of TOPK in cancer patients.