Roberts, Sheryl1
1Institute of Medical and Biological Imaging, Helmholtz Zentrum München, Munich, Germany
________________________________________________________________________
Visualisation of animals for research purposes such as drug discovery and development enables us to aid clinical treatments. Imaging nowadays enables us to non-invasively assess biological structure and function in-vivo with quantitative, spatial and temporal information on normal and diseased models. Imaging is more than just an image. We can quantify physiological function, visualise anatomic detail, characterise whole-body pharmacokinetics, analyse receptor occupancy and the list goes on. In particular, [18F]-fluoro-2-deoxy-D-glucose (FDG) PET is a tomographic imaging technique using a radiolabelled analogue of glucose that can be use to non-invasively visualise tumours and quantify their activity by measuring glucose metabolism. The image and contrast is relative to the glucose uptake rates in various tissues. Coupled with CT is a tomographic technique that uses x-ray beam produces anatomical information, measuring the attenuation coefficients which are converted to Hounsfield-units (H.U.). Combined PET/CT devices are proven diagnostic tools, providing functional information from the [18F]-FDG PET and the anatomical information from the CT in a single examination. Many clinical studies have shown that information obtained by PET/CT yields high accuracy evaluation of coregistration. Here, the effects of varying conditions were analysed retrospectively.
MATERIALS AND METHODS: Dynamic 60 min and static 15 min [18F]-FDG PET/CT data were analysed from two attenuated CT control mouse and compared to two human lymphoma xenografts tumour mouse models. Image analysis and quantification were derived from Amide’s a Medical Image Data Examiner (AMIDE).
| mouse | weight (g) | size of tumour (mm) | fasted/non-fasted | injected dose (Mbq) | anaesthesia(yes/no) | dynamic/static PET |
| (a) control_m1 | 37.1 | n/a | non-fasted | 12.5846 | yes | dynamic, 60 mins |
| (b) control_m2 | 36 | n/a | non-fasted | 11.4025 | no | static,15 mins |
| (c) tumour_m1 | 21.21 | 11.013 | fasted, 2h | 12.9454 | yes | dynamic, 60 mins |
| (d) tumour_m2 | 22.81 | 9.116 | fasted, 2h | 12.3759 | yes | static, 15 mins |
FIGURE 1: Mouse [18F]-FDG studies and its varying experimental conditions.
ANIMAL HANDLING AND PREPARATION: Raw data were obtained from an intensive one week hands on preclinical imaging course offered at the Nuclear Medicine, Rechts der Isar TUM, München. All animal handling were performed in accordance with and approved animal licence. Animals were anaesthetised with isoflurane (2% in oxygen, IsoFlo) inhilation. Mice were kept under anaesthesia, with exception to one mouse (mouse number#) which were awake after tracer injection. They were placed in the imaging chamber at room temp. 25 oC. [18F]-FDG was already synthesised and quality control performed by previously described method that is routinely used in the hospital[1]. ß-decay is via positron emission of 1.66 MeV with a half life of 110 mins.
RECONSTRUCTION AND QUANTITATIVE IMAGE ANALYSIS: Imaging was performed using Inveon CT with image data set dimensions of 256 x 256 x 631 and at a given voxel size 0.173213 mm3. They were reconstructed using Filtered Back Projections (FBP) and Ordered-Subsets Expectation Maximisation (OSEM) 3D algorithms without scatter, random coincidence, dead time, decay and attenuation correction. Quantitative image analysis were performed by AMIDE software and manually drawing regions of interest (ROI) over the following: kidney, heart, liver, lung, muscle and bladder and left tumour. Mice were imaged when the tumour was around 11 and 9 mm. Uptake by [18F]-FDG tracer of various organs is quantified as standardised uptake values (SUVs) using the formula: [activity]tissue (MBq/mL)/injected dose (Bq) x body weight (g).
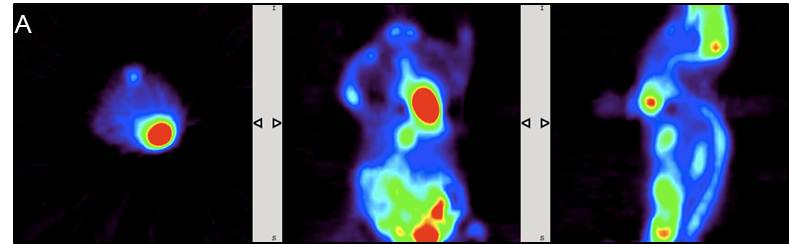
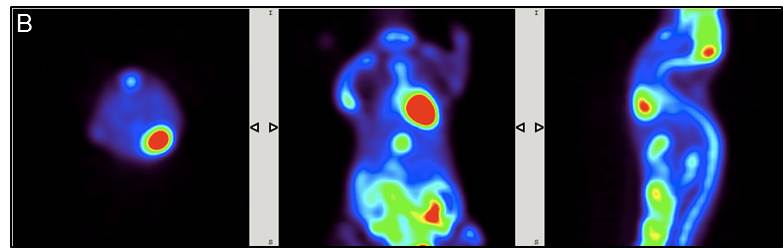
EVALUATION AND RESULTS: [18F]-FDG is a glucose analogue that is readily taken up in cells with high-glucose uptake such as in brain, kidneys and cancer cells where phosphorylation event by hexokinase occurs and prevents the compound to diffuse out of the cell. The biodistribution of [18F]-FDG PET administered isoflurane anaesthetised mouse shows higher activity conc. in the liver, heart, kidneys and bladder as compared to an awake animal at 60 min time-point, perhaps showing quicker blood clearance.
Attenuated Low-med CT scan without contrast agent (CA) [18F]-FDG shows less anatomical information as compared to Low-med CT with CA, showing higher contrast between varying tissue organs.
SUVs showing quicker clearance of [18F]-FDG tracer into the kidney and bladder for tumour_m1 model as compared to control_m1 mouse. The variation greatly depends on the physiological state of the animal.
Image reconstruction with OSEM 3D have a superior image quality in comparison to FBP (below).
Difference in image quality is due to the fact that FBP doesn’t start from 0 histogram which causes “halo” effect and blurring in the reconstruction whereas OSEM-3D has a reference of 0 when back projections occurs.
CONCLUSIONS: Imaging tumour xenografts with 18F-FDG PET/CT with standard procedure were successfully carried out. Administered isoflurane anaesthetised mouse shows higher activity concentration in the liver, heart, kidneys and bladder as compared to an awake animal. Low-med CT scan with contrast agent (CA) [18F]-FDG shows much greater anatomical information than without CA. The magnitude of noise at a pixel level varies in FBP and OSEM projections.
ACKNOWLEDGEMENTS: This study was performed within the framework of the Nuclear Medicine, Rechts der Isar, TUM, München. Data used were derived from hands on preclinical course organised by Prof. Ziegler and Dr. Laitinen.
[1] Hamacher, K.H, J Nucl Med February 1, 1986 vol. 27 no. 2 235-238
© Sheryl Roberts, So you think you can grow crystals in a beaker, 2014 copyright