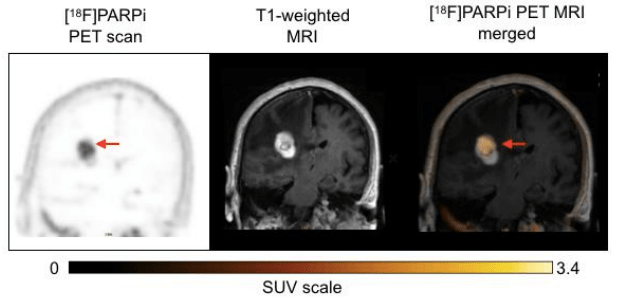
Background: We report preclinical and first-in-human-brain-cancer data using a targeted poly(ADP-ribose)polymerase1 (PARP1) binding PET tracer, [ 18F]PARPi, as a diagnostic tool to differentiate between brain cancers and treatment-related changes.
Methods: We applied a glioma model in p53-deficient nestin/tv-a mice, which were injected with [ 18F]PARPi and then sacrificed 1 hr post-injection for brain examination. We also prospectively enrolled patients with brain cancers to undergo dynamic [ 18F]PARPi acquisition on a dedicated PET/MR scanner. Lesion diagnosis was established by pathology when available or by RANO or RANO-BM response criteria. Resected tissue also underwent PARPi-FL staining and PARP1 immunohistochemistry.
Results: In a preclinical mouse model, we illustrated that [ 18F]PARPi crossed the blood-brain barrier and specifically bound to PARP1 overexpressed in cancer cell nuclei. In humans, we demonstrated high [ 18F]PARPi uptake on PET/MR in active brain cancers and low uptake in treatment-related changes independent of blood-brain barrier disruption. Immunohistochemistry results confirmed higher PARP1 expression in cancerous than in non-cancerous tissue. Specificity was also corroborated by blocking fluorescent tracer uptake with excess unlabeled PARP inhibitor in patient cancer biospecimen.
Conclusions: Although larger studies are necessary to confirm and further explore this tracer, we describe the promising performance of [ 18F]PARPi as a diagnostic tool to evaluate patients with brain cancers and possible treatment-related changes.
