Open until filled.
Check it out our page for updates:
Open until filled.
Check it out our page for updates:
Jan: Transport and relocate equipments and materials & reagents
Feb: Relocated to Yale
March: Setting up lab in new environment
Dr. Roberts is officially a WE AIM Fellow and completed six months leadership training. Oh so many books we read…
Wonderful program from the The Wayne State Empowerment for Academicians in Medicine (WE AIM) focuses on empowering junior and mid‐ career level faculty of the Wayne State University School of Medicine and equipping us with the skills necessary for success within academic medicine. The program is run by The Office of Student Support Services and The Office of Faculty Affairs & Professional Development.
https://facaffairs.med.wayne.edu/weaim


September – Anchorage, Alaska at the World Molecular Imaging Conference
Dr Riya Mallik and Dr. Saheed Ayodeji delivering oral presentations on our work on Estrogen Receptor and Androgen Receptor imaging.
Congrats to Saheed Ayodeji for his 🏆 Travel Award.
Congrats Riya Mallik for her Awards:
🏆 Travel Award,
🏆 Women in Molecular Imaging Network (WIMIN) Scholar Award,
🏆 featured in highlight lecture, and
🏆 Highest scoring abstract from USA 🇱🇷
A well-deserved and a clear testament to both of their work!! We are happy that World Molecular Imaging Society recognized it as well 🎉 It’s been fun and feel privileged to continue working on this together. “Fall seven times, stand up eight” 💪 🇱🇷







With the lab furnished with basic equipments and research supplies, we are open for business!
Mar: Dr. Sharma joined the lab
Sept: Ms. Unaegbu joined the lab
Sept: Dr. Roberts presented at the World Molecular Imaging Conference (WMIC), Prague, Czech Republic
Oct: Dr. Ayodeji joined the lab
Nov: Ms. McColl joined the lab







2022 is the year Sheryl starts from a blank canvas and get the lab up and running…
Institutional Animal and Care and Use Committee (IACUC) protocol approved
Radiation Safety Committee (RSC) protocol approved









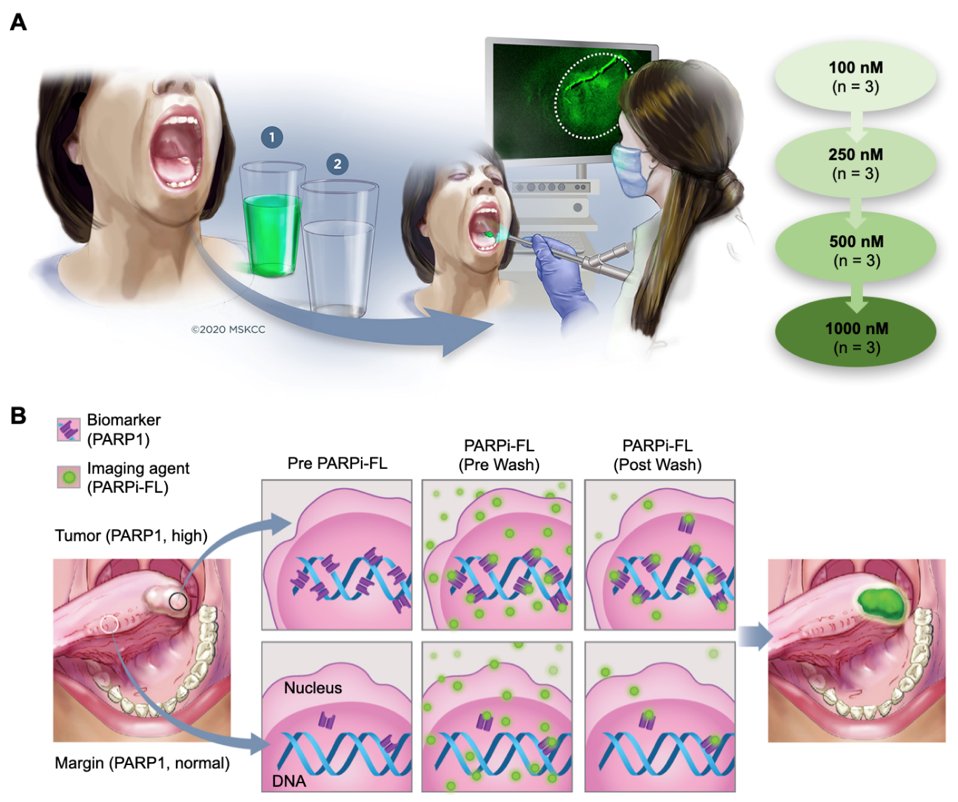
Purpose Visual inspection and biopsy is the current standard of care for oral cancer diagnosis, but is subject to misinterpretation and consequently to misdiagnosis. Topically applied PARPi-FL is a molecularly specific, fluorescent contrast-based approach that may fulfil the unmet need for a simple, in vivo, non-invasive, cost-effective, point-of-care method for the early diagnosis of oral cancer. Here, we present results from a phase I safety and feasibility study on fluorescent, topically applied PARPi-FL.
Translational Relevance Despite their accessible location, oral cavity cancers are often diagnosed late, especially in low-resource areas where their incidence is typically high. The high prevalence of premalignant and benign oral lesions in these populations contributes to a number of issues that make early detection of oral cancer difficult: even in experienced hands, it can be difficult to differentiate cancer from premalignant or benign lesions during routine clinical examination; and biopsy-based histopathology, the current standard of care, is invasive, prone to sampling error, and requires geographic access to appropriate health care professionals, including a highly trained pathologist. While seemingly impenetrable economic and infrastructure barriers have confounded the early diagnosis of oral cancer for most of the world’s population, these could be circumvented by a simple, in vivo, non-invasive, cost-effective, point-of-care method of diagnosis. We are attempting to address this unmet clinical need by using topically applied PARPi-FL — a molecularly specific, fluorescent contrast-based approach — to detect oral cancer.

Limitations in current imaging tools have long challenged the imaging of small pancreatic islets in animal models. Here, we report the first development and in vivo validation testing of a broad spectrum and high absorbance near infrared optoacoustic contrast agent, E4x12-Cy7. Our near infrared tracer (E4x12-Cy7) is based on the amino acid sequence of exendin-4 and targets the glucagon-like peptide-1 receptor (GLP-1R). Cell assays confirmed that E4x12-Cy7 has a high binding affinity (IC50 = 4.6 ± 0.8 nM). Using the multi-spectral optoacoustic tomography (MSOT), we imaged E4x12-Cy7 and optoacoustically visualized β-cell insulinoma xenografts in vivo for the first time. In the future, similar optoacoustic tracers that are specific for β-cells and combines optoacoustic and fluorescence imaging modalities could prove to be important tools for monitoring the pancreas for the progression of diabetes.
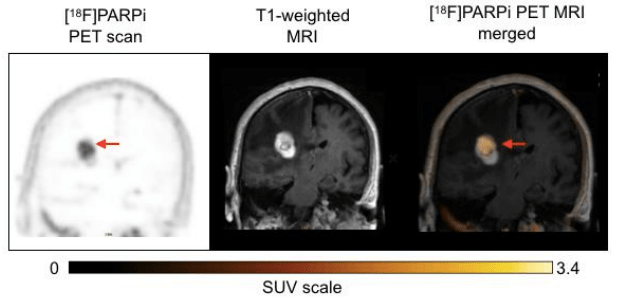
Background: We report preclinical and first-in-human-brain-cancer data using a targeted poly(ADP-ribose)polymerase1 (PARP1) binding PET tracer, [ 18F]PARPi, as a diagnostic tool to differentiate between brain cancers and treatment-related changes.
Methods: We applied a glioma model in p53-deficient nestin/tv-a mice, which were injected with [ 18F]PARPi and then sacrificed 1 hr post-injection for brain examination. We also prospectively enrolled patients with brain cancers to undergo dynamic [ 18F]PARPi acquisition on a dedicated PET/MR scanner. Lesion diagnosis was established by pathology when available or by RANO or RANO-BM response criteria. Resected tissue also underwent PARPi-FL staining and PARP1 immunohistochemistry.
Results: In a preclinical mouse model, we illustrated that [ 18F]PARPi crossed the blood-brain barrier and specifically bound to PARP1 overexpressed in cancer cell nuclei. In humans, we demonstrated high [ 18F]PARPi uptake on PET/MR in active brain cancers and low uptake in treatment-related changes independent of blood-brain barrier disruption. Immunohistochemistry results confirmed higher PARP1 expression in cancerous than in non-cancerous tissue. Specificity was also corroborated by blocking fluorescent tracer uptake with excess unlabeled PARP inhibitor in patient cancer biospecimen.
Conclusions: Although larger studies are necessary to confirm and further explore this tracer, we describe the promising performance of [ 18F]PARPi as a diagnostic tool to evaluate patients with brain cancers and possible treatment-related changes.
Giacomo Pirovano , Sheryl Roberts , Susanne Kossatz and Thomas Reiner*
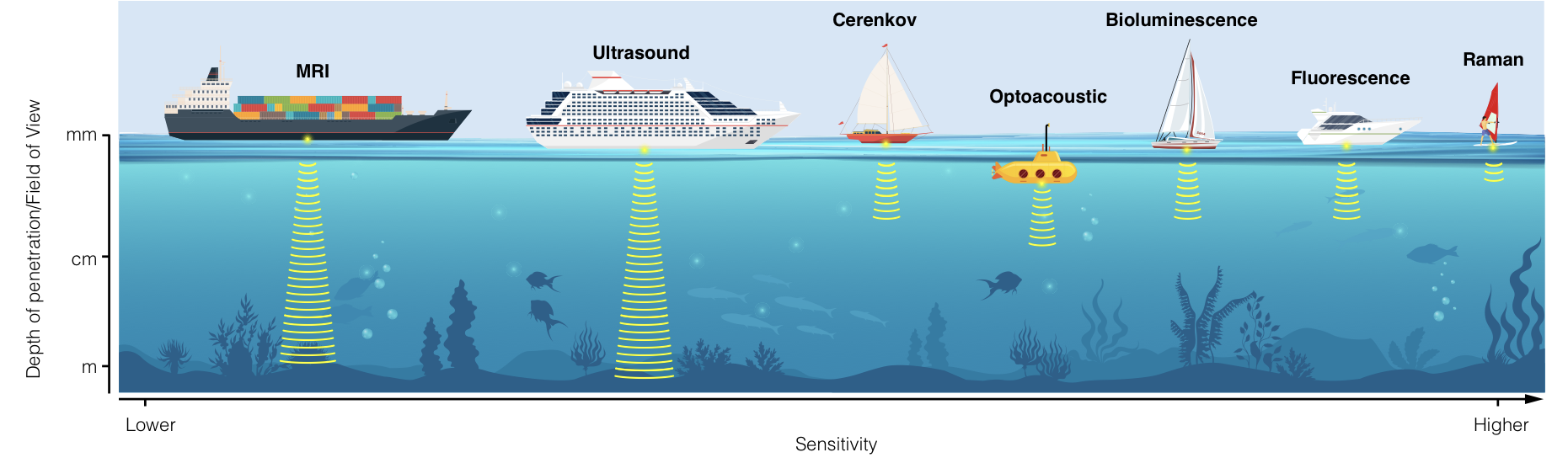
With the ability to non-invasively image and monitor molecular processes within tumors, molecular imaging represents a fundamental tool for cancer scientists. In the current review, we describe emergent optical technologies for molecular imaging. We aim to provide the reader with an overview of the fundamental principles on which each imaging strategy is based, to introduce established and future applications, and to provide a rationale for selecting optical technologies for molecular imaging depending on disease location, biology, and anatomy. In order to accelerate clinical translation of imaging techniques, we also describe examples of practical applications in patients. Elevating these techniques into standard-of-care tools will transform patient stratification, disease monitoring and response evaluation.